By HVN’s Chief Scientist, Professor Richard Mithen
In a few wet gullies within Auckland one can find pukatea trees. I was fortunate to come across some relatively modest specimens recently. They had not yet developed the characteristic buttress roots that will support the mature trees in the decades to come – if these soggy havens of biodiversity are allowed to remain hidden within Auckland suburbia.
Pukatea (Laurelia novae-zealandia) belongs to the plant family Atherospermataceae. It is the only member of the family to occur in New Zealand, with other members being found in South America and Australia.
The bark of pukatea has traditionally been used by Māori as a therapeutic agent for a range of ailments, especially for pain relief. The alkaloid pukateine was isolated in 1909 by Bernard Aston and has some similarities with morphine. Aston was one of New Zealand’s first organic analytic chemists, working within the Department of Agriculture in Wellington, and an accomplished field botanist and conservationist.
Pukateine itself does not seem to have been subject to any thorough investigation of its biological activity, although there is an interesting report suggesting it may enhance dopamine signaling in the brain, which may be of interest concerning therapeutic strategies for Parkinson’s disease.[1]

Pukatea (Laurelia novae-zealandia) and the chemical structure of pukateine, which may be responsible for the traditional use of pukatea bark as an analgesic by Māori.
Barks of trees have been used as therapeutic agents by traditional cultures throughout the world. The stories of the two that have had the most profound effect on our lives are inter-twined – that of the South American Cinchona trees, which contain the anti-malarial alkaloid quinine, and the European Salix trees (willows), which contain the compound salicin from which aspirin was derived.
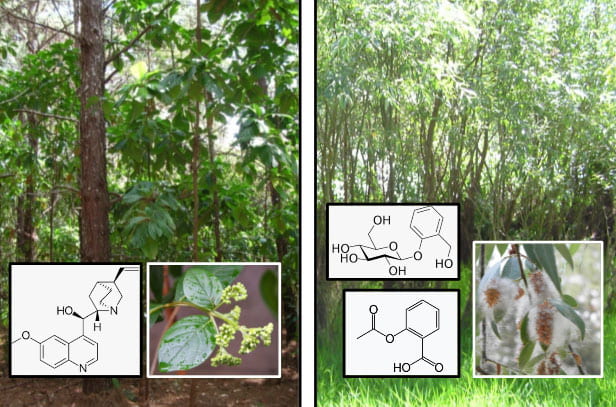
Left: Cinchona pubescens and the structure of quinine. Right: Salix alba and the structure of salicin (above) and its derivative aspirin (below).
Trees of the genus Cinchona[2] are found in the tropical forests of the Andean foothills. It is likely that the bark of Cinchona had a history of use by Quechua and the other native peoples of present-day Peru, Bolivia and Ecuador, but not for malaria, which was brought by European invaders, explorers and missionaries of the 15th Century, and more latterly by enslaved Africans.[3]
The therapeutic effects of cinchona bark to alleviate malaria became known to Spanish Jesuit missionaries in the 16th Century, and it was taken to Europe in the early 17th Century. At the time, malaria was much more prevalent in Europe than it is now. ‘Peruvian bark’ or ‘Jesuit Bark’ became highly sought after, leading to the loss of Cinchona trees in their native habitats.
In due course, crude cinchona bark extracts and quinine itself, after its purification in 1820, enabled Europeans to colonise areas of tropics in which malaria was prevalent, including West and North Africa, Southern India and Southeast Asia. Along with citrus to combat scurvy, quinine would have accompanied European explorers on their first visits to Aotearoa New Zealand in the 18th Century.
Inevitably, demand outstripped the supply of cinchona bark from the forests of South America and ‘fever tree’ plantations were established in Southern India by the British and in Indonesia by the Dutch in the mid-19th Century. It remained the only treatment for malaria until the 1920s, when it was replaced by more effective drugs such as chloroquine. It still retains a place for treatment of mild malaria, especially in areas of resistance to current drugs.
Cinchona bark and quinine are bitter; the Quechua are alleged to have mixed their cinchona bark extracts with sweetened water to make it more palatable, whereas the British used alcohol and invented the ‘Gin and Tonic’.
Whilst there is documentary evidence that extends as far back as 3500 BP[4] for the use of willow as a therapeutic agent, its more recent history starts with a letter written by the Reverend Edward Stone, a Church of England cleric, to the president of the Royal Society in 1763. Stone described how he accidentally tasted some willow bark and found its bitterness reminded him of ‘Peruvian Bark’ (Figure 3).
In the letter, published in the Philosophical Transactions of the Royal Society[5], Reverend Stone describes a series of experiments, firstly upon himself and then upon others over a period of five years, and the effectiveness and safety of willow bark at curing fevers and other disorders. The account is notable for the details provided for the preparation of the willow bark extract, how it was used, and the author’s declaration that his only motive was to give willow bark a “full and fair trial … so the world may reap the benefits accruing from it”. It has the hallmarks of a contemporary scientific publication.

The opening paragraphs of Reverend Edward Stone’s paper on the therapeutic effects of willow bark. Note: His name was incorrectly written as Edmund.
Willow bark and its main active ingredient, salicin, first isolated by the German pharmacologist Johann Buchner in 1828, was widely used as a cheaper and more available substitute for Peruvian bark. It gradually fell out of favour until the Scottish physician Thomas Maclagan, again inspired by the therapeutic properties of cinchona, published a series of clinical trials with salicin in The Lancet. The trials were firstly undertaken upon himself in a manner that we would now term a dose escalation trial, and subsequently on a series of patients with rheumatic fever. [6]
Despite its effectiveness in reducing fever, salicin was not widely adopted as a result of gastric irritation. This was partly solved by the conversion of the glycoside salicin to salicylic acid and then to acetylsalicylic acid, which we know as aspirin, by Arthur Eichengrün and Felix Hoffman in 1897 while working for the German company Bayer.[7] Aspirin became widely used but again fell out of favour due to the development of other analgesics, notably paracetamol in 1956 and ibuprofen in 1962, which lacked the gastric side effects.
Aspirin’s renaissance came through the in-depth studies of its mechanism of action by John Vane and colleagues, which led to the development of nonsteroidal anti-inflammatory drugs (NSAIDs) and cyclooxygenase inhibitors that are widely used today, and the studies by Bengt Samuelsson and colleagues that characterised aspirin’s anti-platelet activity, which led to the current widespread use of aspirin for the prevention of cardiovascular disease. Vane and Samuelssom were awarded the 1982 Nobel Prize for Medicine and Physiology. [8]
Quinine and aspirin, originally derived from the barks of Cinchona and Salix trees, have had a profound effect on global history and medicine. Undoubtedly there are other trees, such as pukatea, that contain natural products that remain to be explored. However, it is likely and maybe appropriate that these trees will keep their secrets deep within the wet gullies and forests of Aotearoa for maybe another era of human history.
[1] Dajas-Bailador et al (1999). Dopaminergic pharmacology and antioxidant properties of pukateine, a natural product lead for the design of agents increasing dopamine neurotransmission. General Pharmacology 32 373-379 DOI: 10.1016/s0306-3623(98)00210-9
[2] Cinchona is a genus of about 23 species bellowing to the Rubiacae. All are native to the tropical forests of South America.
[3] Malaria is caused by species of the single cell microorganism Plasmodium, which is transmitted through mosquitos. There is some limited evidence that one of these species P.vivax may have been present in pre-Colombian South America.
[4] The use of Salix is documented in the Ebers Papyrus, an ancient Egyptian record of herbal remedies dated to 3500 BP.
[5] Stone E (1763). An account of the success of the bark of the willow in the cure of the agues. Philosophical Transactions of the Royal Society of London 53 https://royalsocietypublishing.org/doi/epdf/10.1098/rstl.1763.0033
[6] MacLagan T (1876). The treatment of acute rheumatism by salicin. The Lancet 107, 342-343.
[7] The first account of the discovery of aspirin was published in 1934 but without reference to Eichengrün. This was likely due to Eichengrün being Jewish and the rise of Nazism in 1930s Germany. Eichengrün had left Bayer and established a successful industrial chemist company, which he was forced to sell in 1938 and interred in Theresienstadt concentration camp. In 1949, shortly before his death, he wrote an account of his contribution to the discovery of aspirin. Sneader W (2000). The discovery of aspirin: a reappraisal. BMJ 321.
[8] Samuelsson and Vane shared the 1982 Nobel Prize with Sune Bergstrom.